|
If the instrument includes a scanning monochromator, we can program it to move rapidly to an analyte’s desired wavelength, pause to record its emission intensity, and then move to the next analyte’s wavelength. Schematic diagram of an inductively coupled plasma torch.Ītomic emission spectroscopy is ideally suited for a multielemental analysis because all analytes in a sample are excited simultaneously. This is accomplished by the tangential flow of argon shown in the schematic diagram. At these high temperatures the outer quartz tube must be thermally isolated from the plasma. The resulting collisions with the abundant unionized gas give rise to resistive heating, providing temperatures as high as 10000 K at the base of the plasma, and between 60 K at a height of 15–20 mm above the coil, where emission usually is measured. 
An alternating radio-frequency current in the induction coil creates a fluctuating magnetic field that induces the argon ions and the electrons to move in a circular path. Plasma formation is initiated by a spark from a Tesla coil. 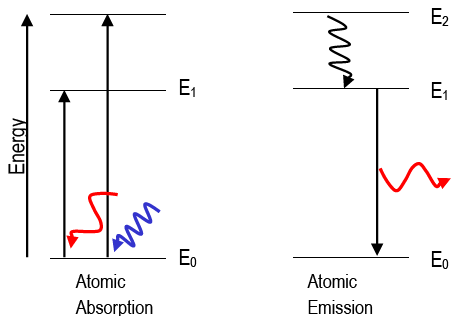
The sample is mixed with a stream of Ar using a nebulizer, and is carried to the plasma through the torch’s central capillary tube. The ICP torch consists of three concentric quartz tubes, surrounded at the top by a radio-frequency induction coil. Because a plasma operates at a much higher temperature than a flame, it provides for a better atomization efficiency and a higher population of excited states.Ī schematic diagram of the inductively coupled plasma source (ICP) is shown in Figure 10.7.2 A plasma’s high temperature results from resistive heating as the electrons and argon ions move through the gas. The plasma used in atomic emission is formed by ionizing a flowing stream of argon gas, producing argon ions and electrons. We also expect emission intensity to increase with temperature.Ī plasma is a hot, partially ionized gas that contains an abundant concentration of cations and electrons. I would encourage you to explore further.We expect that excited states with lower energies have larger populations and more intense emission lines. 
looking for relationships in a meaningful way. We can, however, say that the more electrons there are in an atom, the greater the variety of photon energies there will be so this may be a link worth exploringĪs I say, you are asking a great question. as it switches from one energy level to another, then it emits / absorbs photons. This is because the electron can exist in many energy levels. Hydrogen, for example, although the simplest atom, has a whole range of photon energies that it emits. The value of the energies of photons does depend on the available energy levels in the atom. Now, the energy of the photon emitted from any element does not depend on the number of electrons in the atom. OK: I would say that the periodic table tells us about the number of protons in an element and, therefore the number of electrons too. Here is my answer, but I would encourage you to explore this and similar questions further. Planck studied the electromagnetic radiation emitted by heated objects, and he proposed that the emitted electromagnetic radiation was "quantized" since the energy of light could only have values given by the following equation: E photon = n h ν E_ s 1 start fraction, 1, divided by, start text, s, end text, end fraction. 
Physicists Max Planck and Albert Einstein had recently theorized that electromagnetic radiation not only behaves like a wave, but also sometimes like particles called photons. By the early 1900s, scientists were aware that some phenomena occurred in a discrete, as opposed to continuous, manner.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
